U.S. Ophthalmic Devices And Eye Care Market
U.S. Ophthalmic Devices And Eye Care Market Size, Share & Trends Analysis Report By Product (Ophthalmic Devices, Eye Care), By Application (Cataract, Glaucoma, Refractive Disorders, Vitreo Retinal Disorders), By End-use, And Segment Forecasts
Published Date: August - 2025 | Publisher: MIR | No of Pages: 250 | Industry: helthcare | Format: Report available in PDF / Excel Format
View Details Download Free Sample Ask for Discount Request CustomizationClaim a Free Sample
Get a preview with key statistics, projections, and trends for this report. Ideal for quick insights.
Send Me a SampleNeed full access?
Purchase ReportMarket Size & Trends
The market for eye care and ophthalmic devices in the United States was valued at USD 22.9 billion in 2024 and is expected to expand at a compound annual growth rate (CAGR) of 7.14% between 2025 and 2030.
![]()
U.S. Ophthalmic Devices And Eye Care Market
Due to the growing number of elderly people who are more vulnerable to eye problems such cataracts, glaucoma, diabetic retinopathy, and refractive errors, the U.S. market is expanding. The CDC WONDER, Single-Race Population Estimates, 2023, for example, estimates that 17.7% of the nation's population was 65 years of age or older in 2023. The market is also expanding as a result of strategic industry initiatives such creative product launches and partnerships, as well as rising awareness of eye health.A primary driver for the market’s growth is the rapidly aging population. Individuals aged 65 and older are disproportionately affected by age-related eye conditions such as cataracts, age-related macular degeneration (AMD), glaucoma, and presbyopia, necessitating increased demand for diagnostic services, surgical interventions, and vision correction. In 2022, an estimated 4.22 million US adults (1.62%) had glaucoma, and 1.49 million (0.57%) had vision-affecting glaucoma, according to data published by the CDC in October 2024. Similarly, the rising prevalence of systemic diseases such as diabetes contributes significantly to the burden of eye disease, particularly diabetic retinopathy, further boosting the need for specialized eye care and related devices. According to the International Diabetes Federation, the prevalence of diabetes in the U.S. is estimated to be around 13.7% in 2024, with approximately 38.5 million people living with the disease.
The U.S. ophthalmic devices and eye care industry is characterized by increasing investments, a supportive regulatory framework, a growing focus on innovation, and an evolving competitive landscape. Moreover, the high prevalence of health disorders and an aging population in the country significantly increase the growth opportunities for the market.
The high degree of innovation in the U.S. ophthalmic devices and eye care industry is driven by advancements in technology and increasing demand for effective vision correction solutions. The integration of artificial intelligence (AI) in diagnostic tools has revolutionized how conditions such as diabetic retinopathy are detected, allowing for earlier intervention and improved patient outcomes. For instance, in May 2024, AEYE Health received FDA clearance for the first fully autonomous AI to diagnose diabetic retinopathy using handheld retinal cameras. The system demonstrated high sensitivity and specificity in trials and requires only one image per eye. It enables point-of-care, reimbursed screenings without dilation, enhancing accessibility and potentially preventing blindness. Furthermore, the rise of telemedicine platforms has expanded access to eye care services, enabling remote consultations and monitoring, which is significantly beneficial in underserved areas.
The U.S. ophthalmic devices and eye care industry has witnessed a significant rise in mergers and acquisitions (M&A) activities, driven by technological advancements and an increasing demand for innovative solutions. For instance, in August 2024, Advancing Eyecare (AEC) acquired Eyefficient, expanding its ophthalmic equipment offerings and nationwide reach. Eyefficient, known for quality, affordable diagnostic instruments, will leverage AEC’s infrastructure to enhance customer support. This partnership strengthens AEC’s offerings in eye eyecare instrumentation, benefiting distributors and independent sales representatives with broader product access and services. The M&A landscape is also shaped by strategic partnerships that aim to address evolving consumer needs while enhancing competitive positioning within the market.
The regulatory landscape for U.S. ophthalmic devices and the eye care industry is primarily governed by the Food and Drug Administration (FDA), which classifies these devices into three categories based on riskClass I, II, and III. This classification impacts market entry, as Class III devices require premarket approval (PMA), a process that can be lengthy and costly due to extensive clinical trials and data requirements. In addition, regulations surrounding reimbursement policies from Medicare and private insurers significantly influence market dynamics; devices that demonstrate clear clinical benefits are more likely to receive favorable reimbursement rates. Thus, companies need to manage regulatory hurdles and market access challenges to launch new products successfully in this competitive sector.
The U.S. ophthalmic devices and eye care industry faces significant threats from substitutes as advancements in technology and consumer preferences evolve. The increasing use of telemedicine for eye care consultations, which allows patients to receive assessments and prescriptions without traditional in-person visits, offers a substitute for these devices, as this shift reduces the demand for conventional eye examinations and encourages the adoption of home-based diagnostic tools, such as smartphone apps that can assess vision quality or detect common ocular conditions.
The U.S. ophthalmic devices and eye care industry is witnessing significant regional expansion, driven by increasing demand for advanced eye care solutions and a growing aging population. Companies are strategically enhancing their presence across various states to address the increasing demand for ophthalmic devices and eye care solutions. For instance, in March 2023, CooperVision announced that the company had increased the delivery times of Biofinity toric multifocal contact lenses by over 50% for U.S. and Canadian customers after moving the manufacturing to its advanced facility in Scottsville, New York, in late 2022. These made-to-order lenses ship within approximately one week, enhancing patient care with faster follow-ups and prescription adjustments. This move improves accessibility and satisfaction for eye care professionals and patients. As competition intensifies, these companies are expected to continue expanding their footprint across the U.S., aiming to capture a larger share of the market.
Product Insights
The eye care segment dominated the market with the largest revenue share of around 55.7% in 2024, owing to consistent demand for corrective and therapeutic products, especially contact lenses and intraocular lenses (IOLs). The contact lens demand is fueled by a large base of myopic and astigmatic patients, particularly in younger, working-aged populations seeking alternatives to eyeglasses for aesthetic and functional reasons. Similarly, intraocular lenses witness demand owing to the high volume of cataract surgeries, one of the most frequently performed procedures in the U.S. Compared to diagnostic or surgical ophthalmic devices, eye care products are consumables with repetitive usage patterns, which increases their demand in the market.
The ophthalmic devices segment is expected to witness the fastest CAGR of 7.60% over the forecast period, driven by rising demand for advanced diagnostic and surgical instruments. Age-related eye conditions, including cataracts, glaucoma, and age-related macular degeneration (AMD), are increasingly prevalent and drive the demand for precision equipment like optical coherence tomography (OCT) systems and fundus cameras. Additionally, the growing use of AI for analyzing images to spot diabetic retinopathy and glaucoma early in eye care devices is also driving this growth. For instance, in January 2025, AI Optics’ portable Sentinel Camera received FDA 510(k) clearance. This handheld retinal imaging device captures high-quality images without eye dilation, integrates with EHR systems, and aims to improve retinal disease screening accessibility.
Report Coverage & Deliverables
The PDF report and online dashboard will help you understand
- Competitive benchmarking
- Historical data & forecasts
- Company revenue shares
- Regional opportunities
- Latest trends & dynamics
Application Insights
Cataracts were the most common reason for procedures in 2024 and are likely to grow the fastest because many older people have age-related cataracts, leading to a high demand for treatments and devices. An estimated 20.5 million Americans aged 40 and older, representing 17.2% of this population, have cataracts in one or both eyes, and approximately 6.1 million (5.1%) have undergone surgery to remove their lens, according to CDC data from May 2024. The high prevalence of cataracts among the population contributes to growth in this market segment.
Glaucoma is expected to witness significant CAGR owing to the expanding adoption of advanced diagnostic and surgical technologies. The growing use of tools like optical coherence tomography (OCT) and tonometers for spotting glaucoma early, especially in outpatient surgical centers and specialized eye clinics, is driving up demand. Using advanced eye devices provides safer and more effective long-term control of eye pressure with fewer complications, which fits well with the move towards better care and treatments that can be done outside of hospitals.
End-use Insights
The hospitals and eye clinics segment dominated the market with the largest revenue share of around 77.4% in 2024, owing to their crucial role in the diagnosis, treatment, and surgical management of complex ocular conditions, including cataracts, glaucoma, and retinal disorders. These facilities are the primary sites for high-volume procedures such as phacoemulsification cataract surgeries and intravitreal injections, which require advanced ophthalmic devices. The widespread availability of specialized ophthalmologists in these settings, combined with access to reimbursement through Medicare and private insurance for procedures performed in accredited facilities, further drives segment growth.
The academic and research laboratory sector is likely to grow a lot because more money is being put into vision science and there are more complicated eye diseases that need better ways to diagnose and treat them. These laboratories are leading the way in creating and testing new technologies like adaptive optics, optical coherence tomography (OCT), and AI-assisted retinal imaging, which are essential for spotting diseases early. In addition, partnerships between academic institutions and med-tech companies have accelerated prototype testing and clinical validation, increasing demand for advanced ophthalmic devices in lab environments.
Key U.S. Ophthalmic Devices And Eye Care Company Insights
Some of the leading market players include Nidek Co. Ltd., Johnson & Johnson Vision Care, Sevenoe, Duckworth & Kent, and Alcon, among others. These companies have utilized their technological expertise to establish a significant presence in this growing market. Their strategies include user-friendly solutions, emphasis on affordability, and improved accessibility, which ensure widespread adoption of their advanced products.
Key U.S. Ophthalmic Devices And Eye Care Companies
- Nidek Co. Ltd.
- Johnson & Johnson Vision Care
- Sevenoe
- Duckworth& Kent
- Alcon
- Carl Zeiss Meditec AG
- Topcon Corporation
- NIDEK Co., Ltd.
- Glaukos Corporation
- CooperVision, Inc.
Recent Developments
-
In October4, Visionix launched the VX 610, a nonmydriatic automated fundus camera requiring only a 2-mm pupil and no dilation, using cross-polarized light. It features auto alignment, tablet control, a 45-degree field of view, and 90-degree mosaic imaging. Telehealth-ready, it supports diabetic retinopathy screening and integrates with EMRs.
-
In May 2024, Reichert Technologies announced the availability of the Reichert Tono-Vera Tonometer in the U.S. It offers objective, accurate intraocular pressure measurement with an ergonomic design and digital alignment. This device aims to improve glaucoma screening and management by providing reliable results and enhancing patient comfort during eye examinations.
-
In January4, Nidek Inc. commercially launched the Mirante Scanning Laser Ophthalmoscope (SLO) in the U.S. This device combines high-quality SLO and OCT with wide-angle imaging, allowing it to take clear color images, indocyanine green angiography, fluorescein angiography, Retro mode, fundus autofluorescence, and OCT images all at once. The device supports dynamic blood flow imaging and detailed retinal structure evaluation, enhancing diagnosis and management of retinal disease.
U.S. Ophthalmic Devices And Eye Care Market Report Scope
|
Report Attribute |
Details |
|
Market size value in 2025 |
USD 24.29 billion |
|
Revenue forecast in 2030 |
USD 34.29 billion |
|
Growth rate |
CAGR of 7.14% from 2025 to 2030 |
|
Actual Data |
2018-2024 |
|
Forecast period |
2025-2030 |
|
Quantitative units |
Revenue in USD million/billion, and CAGR from 2025 to 2030 |
|
Report coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
|
Segments covered |
Product, application, end-use |
|
Regional scope |
U.S. |
|
Key companies profiled |
Nidek Co., Ltd.; Johnson & Johnson Vision Care; Sevenoe; Duckworth & Kent; Alcon; Carl Zeiss Meditec AG; Topcon Corporation; NIDEK Co., Ltd.; Glaukos Corporation; CooperVision, Inc. |
|
Customization scope |
Free report customization (equivalent to up to 8 analysts' working days) with purchase. You can add or alter the segment scope. |
|
Pricing and purchase options |
Avail yourself of customized purchase options to meet your exact research needs. Explore purchase options |
U.S. Ophthalmic Devices And Eye Care Market Report Segmentation
This report forecasts revenue growth at the country level and analyzes the latest industry trends in each of the subsegments from 2018 to 2030. For this study, Grand View Research has segmented the U.S. ophthalmic devices and eye care market report based on product, application, and end-use
-
Product Outlook (Revenue, USD Million, 2018-2030)
-
Ophthalmic Devices
-
Optical Coherence Tomography Scanners
-
Fundus Cameras
-
Perimeters/Visual Field Analyzers
-
Autorefractors and Keratometers
-
Ophthalmic Ultrasound Imaging Systems
-
Tonometers
-
Slit Lamps
-
Phoropters
-
Wavefront Aberrometers
-
Optical Biometry Systems
-
Ophthalmoscopes
-
Lensmeters
-
Corneal Topography Systems
-
Specular Microscopes
-
Retinoscopes
-
-
Eye Care
-
Contact Lenses
-
Intraocular Lenses
-
Ocular Health Products
-
Others
-
-
-
Application Outlook (Revenue, USD Million, 2018-2030)
-
Cataract
-
retinal disorders
-
Glaucoma
-
Refractive Disorders
-
-
End-use Outlook (Revenue, USD Million, 2018-2030)
-
Hospitals and Eye Clinics
-
Academic and Research Laboratory
-
Others
-
Frequently Asked Questions
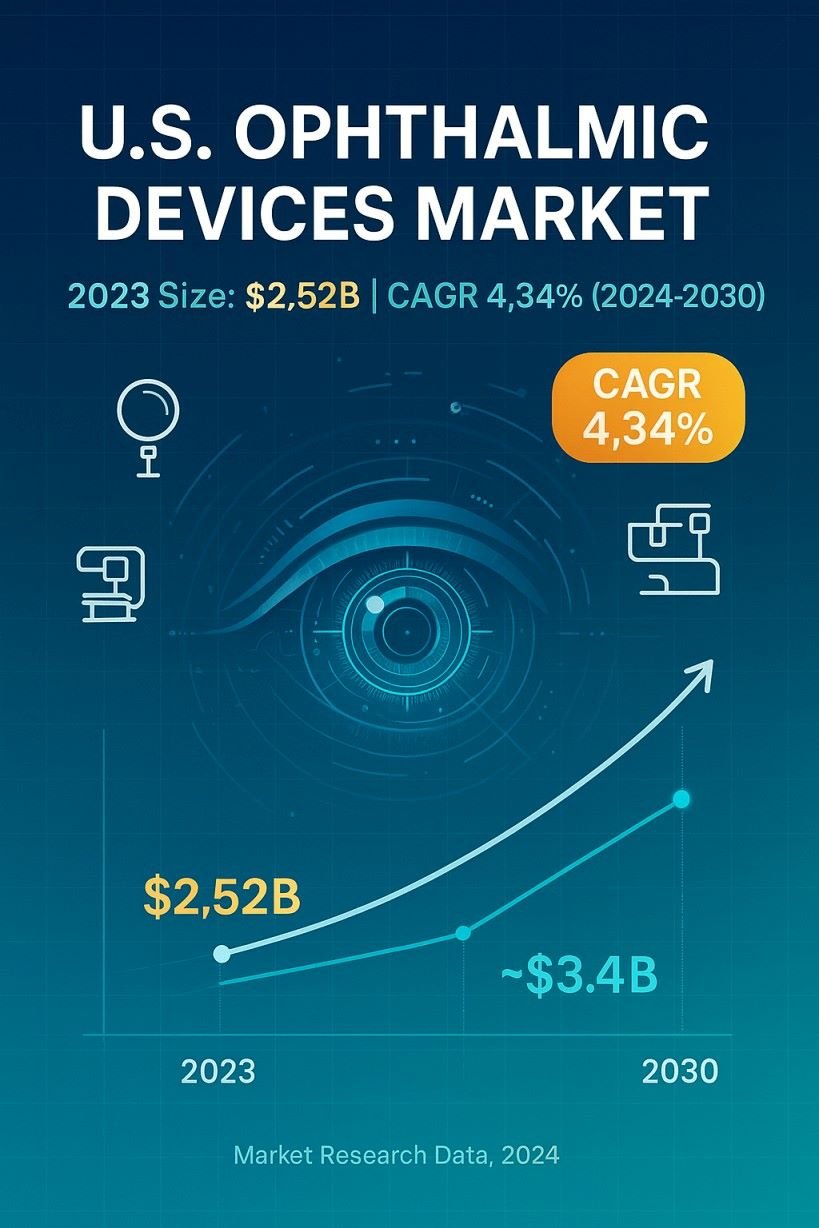
The U.S. ophthalmic devices market size was estimated at USD 2.52 billion in 2023 and is expected to reach USD 2.65 billion in 2024.
The U.S. ophthalmic devices market is expected to grow at a compound annual growth rate of 4.3% from 2024 to 2030 to reach USD 3.40 billion by 2030.
Based on product, optical coherence tomography scanners dominated the ophthalmic devices market with a share of 23.2% in 2023. This is attributable to advancements in OCT, which help in the efficient diagnosis and treatment of retinal disorders, glaucoma, and other ophthalmic disorders.
b.Some key players operating in the U.S. ophthalmic devices market include Abbott Medical Optics, Inc., Carl Zeiss Meditec AG, Alcon, Inc., Bausch & Lomb, Inc., Haag-Streit, Topcon Corporation, Johnson & Johnson, Essilor International S.A., Ziemer Ophthalmic Systems AG, and Nidek Co. Ltd.Some key players operating in the U.S. ophthalmic devices market include Abbott Medical Optics, Inc., Carl Zeiss Meditec AG, Alcon, Inc., Bausch & Lomb, Inc., Haag-Streit, Topcon Corporation, Johnson & Johnson, Essilor International S.A., Ziemer Ophthalmic Systems AG, and Nidek Co. Ltd
Key factors that are driving the U.S. ophthalmic devices market growth include the rising prevalence of eye diseases, technological advancements in ophthalmic devices, and government initiatives for increasing awareness regarding visual impairment.
Related Reports
- Sterile Bioprocess Filtration Market Size, Share & Trends Analysis Report By Product (Membrane Filters, Cartridge Filter...
- Computational Pathology Market Size, Share & Trends Analysis Report By Component (Software, Services) By Application, By...
- Non-invasive Brain Trauma Monitoring Device Market Forecast 2025–2035: Revolutionizing Neurological Care with AI, Wear...
- Germany Accident Insurance Market Size (Gross Written Premium, New Business Premium), Share & Trends Analysis Report By ...
- U.S. Surgical Dressings Market Size, Share & Trends Analysis Report By Product (Primary, Secondary), By Application (Ulc...
- High Flow Nasal Cannula Market: Trends and Forecast 2025 to 2035
Table of Content
Table of Contents
Chapter 1. Methodology and Scope
1.1. Market Segmentation & Scope
1.2. Market Definitions
1.2.1. Product segment
1.2.2. Application segment
1.2.3. End Use segment
1.3. Information analysis
1.3.1. Market formulation & data visualization
1.4. Data validation & publishing
1.5. Information Procurement
1.5.1. Primary research
1.6. Information or Data Analysis
1.7. Market Formulation & Validation
1.8. Market Model
1.9. Total Market: CAGR Calculation
1.10. Objectives
1.10.1. Objective 1
1.10.2. Objective 2
Chapter 2. Executive Summary
2.1. Market Outlook
2.2. Segment Snapshot
2.3. Competitive Insights Landscape
Chapter 3. U.S. Ophthalmic Devices and Eye Care Market Variables, Trends & Scope
3.1. Market Lineage Outlook
3.1.1. Parent market outlook
3.1.2. Related/ancillary market outlook.
3.2. Market Dynamics
3.2.1. Market driver analysis
3.2.1.1. Rising prevalence of eye diseases
3.2.1.2. Increasing technological advancements
3.2.1.3. Growing geriatric population
3.2.2. Market restraint analysis
3.2.2.1. High cost of advanced devices
3.2.2.2. Stringent regulatory requirements
3.3. Ophthalmic Devices and Eye Care Market Analysis Tools
3.3.1. Industry analysis - Porter’s
3.3.2. Pestel analysis
Chapter 4. U.S. Ophthalmic Devices and Eye Care Market: Product Estimates & Trend Analysis
4.1. Segment Dashboard
4.2. U.S. Ophthalmic Devices and Eye Care Market Product Movement Analysis
4.3. U.S. Ophthalmic Devices and Eye Care Market Size & Trend Analysis, by Product, 2018 to 2030 (USD Million)
4.4. Ophthalmic Devices
4.4.1. Ophthalmic devices market estimates and forecasts 2018 to 2030 (USD Million)
4.4.2. Optical coherence tomography scanners
4.4.2.1. Optical coherence tomography scanners market estimates and forecasts 2018 to 2030 (USD Million)
4.4.3. Fundus cameras
4.4.3.1. Fundus cameras market estimates and forecasts 2018 to 2030 (USD Million)
4.4.4. Perimeters/visual field analyzers
4.4.4.1. Perimeters/visual field analyzers market estimates and forecasts 2018 to 2030 (USD Million)
4.4.5. Autorefractors and keratometers
4.4.5.1. Autorefractors and keratometers market estimates and forecasts 2018 to 2030 (USD Million)
4.4.6. Ophthalmic ultrasound imaging systems
4.4.6.1. Ophthalmic ultrasound imaging systems market estimates and forecasts 2018 to 2030 (USD Million)
4.4.7. Tonometers
4.4.7.1. Tonometers market estimates and forecasts 2018 to 2030 (USD Million)
4.4.8. Slit lamps
4.4.8.1. Slit lamps market estimates and forecasts 2018 to 2030 (USD Million)
4.4.9. Phoropters
4.4.9.1. Phoropters market estimates and forecasts 2018 to 2030 (USD Million)
4.4.10. Wavefront aberrometers
4.4.10.1. Wavefront aberrometers market estimates and forecasts 2018 to 2030 (USD Million)
4.4.11. Optical biometry systems
4.4.11.1. Optical biometry systems market estimates and forecasts 2018 to 2030 (USD Million)
4.4.12. Ophthalmoscopes
4.4.12.1. Ophthalmoscopes market estimates and forecasts 2018 to 2030 (USD Million)
4.4.13. Lensmeters
4.4.13.1. Lensmeters market estimates and forecasts 2018 to 2030 (USD Million)
4.4.14. Corneal topography systems
4.4.14.1. Corneal topography systems market estimates and forecasts 2018 to 2030 (USD Million)
4.4.15. Specular microscopes
4.4.15.1. Specular microscopes market estimates and forecasts 2018 to 2030 (USD Million)
4.4.16. Retinoscopes
4.4.16.1. Retinoscopes market estimates and forecasts 2018 to 2030 (USD Million)
4.5. Eye Care
4.5.1. Eye care market estimates and forecasts 2018 to 2030 (USD Million)
4.5.2. Contact lenses
4.5.2.1. Contact lenses market estimates and forecasts 2018 to 2030 (USD Million)
4.5.3. Intraocular lenses
4.5.3.1. Intraocular lenses market estimates and forecasts 2018 to 2030 (USD Million)
4.5.4. Ocular health products
4.5.4.1. Ocular health products market estimates and forecasts 2018 to 2030 (USD Million)
4.5.5. Others
4.5.5.1. Others market estimates and forecasts 2018 to 2030 (USD Million)
Chapter 5. U.S. Ophthalmic Devices and Eye Care Market: Application Estimates & Trend Analysis
5.1. Segment Dashboard
5.2. U.S. Ophthalmic Devices and Eye Care Market Application Movement Analysis
5.3. U.S. Ophthalmic Devices and Eye Care Market Size & Trend Analysis, by Application, 2018 to 2030 (USD Million)
5.4. Cataract
5.4.1. Cataract market estimates and forecasts 2018 to 2030 (USD Million)
5.5. Vitreo retinal disorders
5.5.1. Vitreo retinal disorders market estimates and forecasts 2018 to 2030 (USD Million)
5.6. Glaucoma
5.6.1. Glaucoma market estimates and forecasts 2018 to 2030 (USD Million)
5.7. Refractive disorders
5.7.1. Refractive disorders market estimates and forecasts 2018 to 2030 (USD Million)
Chapter 6. U.S. Ophthalmic Devices and Eye Care Market: End Use Estimates & Trend Analysis
6.1. Segment Dashboard
6.2. U.S. Ophthalmic Devices and Eye Care Market End Use Movement Analysis
6.3. U.S. Ophthalmic Devices and Eye Care Market Size & Trend Analysis, by End Use, 2018 to 2030 (USD Million)
6.4. Hospitals and eye clinics
6.4.1. Hospitals and eye clinics market estimates and forecasts 2018 to 2030 (USD Million)
6.5. Academic and research laboratory
6.5.1. Academic and research laboratory market estimates and forecasts 2018 to 2030 (USD Million)
6.6. Others
6.6.1. Others market estimates and forecasts 2018 to 2030 (USD Million)
Chapter 7. Competitive Landscape
7.1. Company/Competition Categorization
7.2. Strategy Mapping
7.3. Company Market Position Analysis, 2024
7.4. Company Profiles/Listing
7.4.1. Nidek Co. Ltd.
7.4.1.1. Company overview
7.4.1.2. Financial performance
7.4.1.3. Product benchmarking
7.4.1.4. Strategic initiatives
7.4.2. Johnson & Johnson Vision Care
7.4.2.1. Company overview
7.4.2.2. Financial performance
7.4.2.3. Product benchmarking
7.4.2.4. Strategic initiatives
7.4.3. Sevenoe
7.4.3.1. Company overview
7.4.3.2. Financial performance
7.4.3.3. Product benchmarking
7.4.3.4. Strategic initiatives
7.4.4. Duckworth& Kent
7.4.4.1. Company overview
7.4.4.2. Financial performance
7.4.4.3. Product benchmarking
7.4.4.4. Strategic initiatives
7.4.5. Alcon
7.4.5.1. Company overview
7.4.5.2. Financial performance
7.4.5.3. Product benchmarking
7.4.5.4. Strategic initiatives
7.4.6. Carl Zeiss Meditec AG
7.4.6.1. Company overview
7.4.6.2. Financial performance
7.4.6.3. Product benchmarking
7.4.6.4. Strategic initiatives
7.4.7. Topcon Corporation
7.4.7.1. Company overview
7.4.7.2. Financial performance
7.4.7.3. Product benchmarking
7.4.7.4. Strategic initiatives
7.4.8. NIDEK Co., Ltd.
7.4.8.1. Company overview
7.4.8.2. Financial performance
7.4.8.3. Product benchmarking
7.4.8.4. Strategic initiatives
7.4.9. Glaukos Corporation
7.4.9.1. Company overview
7.4.9.2. Financial performance
7.4.9.3. Product benchmarking
7.4.9.4. Strategic initiatives
7.4.10. CooperVision, Inc.
7.4.10.1. Company overview
7.4.10.2. Financial performance
7.4.10.3. Product benchmarking
7.4.10.4. Strategic initiatives
List of Tables
Table 1 List of secondary sources
Table 2 List of abbreviations
Table 3 U.S. ophthalmic devices and eye care market by product, 2018 - 2030 (USD Million)
Table 4 U.S ophthalmic devices and eye care market by application, 2018 - 2030 (USD Million)
Table 5 U.S ophthalmic devices and eye care market by end use, 2018 - 2030 (USD Million)
Table 6 Participant’s overview
Table 7 Financial performance
Table 8 Key companies undergoing expansions
Table 9 Key companies undergoing acquisitions
Table 10 Key companies undergoing collaborations
Table 11 Key companies launching new products
Table 12 Key companies undergoing partnerships
Table 13 Key companies undertaking other strategies
List of Figures
Fig. 1 Market research process
Fig. 2 Data triangulation techniques
Fig. 3 Market formulation & validation
Fig. 4 U.S. ophthalmic devices and eye care market: Market outlook
Fig. 5 U.S. ophthalmic devices and eye care market: Segment outlook
Fig. 6 U.S. ophthalmic devices and eye care market: Competitive landscape outlook
Fig. 7 Parent market outlook
Fig. 8 U.S. ophthalmic devices and eye care market driver impact
Fig. 9 U.S. ophthalmic devices and eye care market restraint impact
Fig. 10 U.S. ophthalmic devices and eye care market: Product outlook and key takeaways
Fig. 11 U.S. ophthalmic devices and eye care market: Product movement analysis
Fig. 12 Ophthalmic devices market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 13 Optical coherence tomography scanners market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 14 Fundus cameras market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 15 Perimeters/visual field analyzers market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 16 Autorefractors and keratometers market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 17 Ophthalmic ultrasound imaging systems market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 18 Tonometers market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 19 Slit lamps market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 20 Phoropters market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 21 Wavefront aAberrometers market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 22 Optical biometry systems market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 23 Ophthalmoscopes market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 24 Lensmeters market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 25 Corneal topography systems market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 26 Specular microscopes market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 27 Retinoscopes market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 28 Eye care market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 29 Contact lenses market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 30 Intraocular lenses market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 31 Ocular health products market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 32 Others market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 33 U.S. ophthalmic devices and eye care market: Application outlook and key takeaways
Fig. 34 U.S. ophthalmic devices and eye care market: Application movement analysis
Fig. 35 Cataract market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 36 Vitreo retinal disorders market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 37 Glaucoma market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 38 Refractive disorders market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 39 U.S. ophthalmic devices and eye care market: End Use outlook and key takeaways
Fig. 40 U.S. ophthalmic devices and eye care market: End Use movement analysis
Fig. 41 Hospitals and eye clinics market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 42 Academic and research laboratory market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 43 Others market estimates and forecasts, 2018 - 2030 (USD Million)
Fig. 44 Heat map analysis
Fig. 45 Market participant categorization
Fig. 46 U.S. ophthalmic devices and eye care market share analysis, 2024
Fig. 47 Strategy framework
Key U.S. Ophthalmic Devices And Eye Care Companies:
- Nidek Co. Ltd.
- Johnson & Johnson Vision Care
- Sevenoe
- Duckworth& Kent
- Alcon
- Carl Zeiss Meditec AG
- Topcon Corporation
- NIDEK Co., Ltd.
- Glaukos Corporation
- CooperVision, Inc.
